1. Kario, K. Nocturnal Hypertension. Hypertension 71, 997-1009 (2018).
2. Kario, K. Morning surge in blood pressure and cardiovascular risk: evidence and perspectives. Hypertension 56, 765-773 (2010).
3. Kario, K. et al. The HOPE Asia network 2022 up-date consensus statement on morning hypertension management. Journal of clinical hypertension (Greenwich, Conn.) 24, 1112-1120 (2022).
4. Sharma, T., Mathur, P. & Mehta, J.L. Daytime or Nighttime Administration of Antihypertensive Medications? American journal of hypertension 33, 987-989 (2020).
5. Hermida, R.C., Ayala, D.E., Mojon, A. & Fernandez, J.R. Influence of circadian time of hypertension treatment on cardiovascular risk: results of the MAPEC study. Chronobiol Int 27, 1629-1651 (2010).
6. Hermida, R.C. et al. Bedtime hypertension treatment improves cardiovascular risk reduction: the Hygia Chronotherapy Trial. Eur Heart J 41, 4565-4576 (2020).
7. Stergiou, G. et al. Bedtime dosing of antihypertensive medications: systematic review and consensus statement: International Society of Hypertension position paper endorsed by World Hypertension League and European Society of Hypertension. J Hypertens 40, 1847-1858 (2022).
8. Turgeon, R.D. et al. Lowering Nighttime Blood Pressure With Bedtime Dosing of Antihypertensive Medications: Controversies in Hypertension - Con Side of the Argument. Hypertension 78, 871-878 (2021).
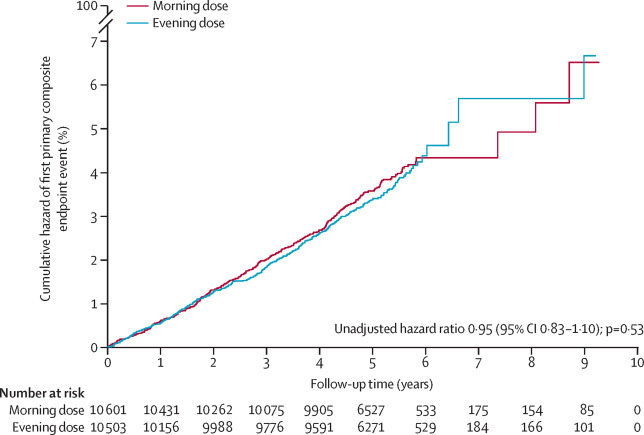
9. Mackenzie, I.S. et al. Cardiovascular outcomes in adults with hypertension with evening versus morning dosing of usual antihypertensives in the UK (TIME study): a prospective, randomised, open-label, blinded-endpoint clinical trial. Lancet (London, England) 400, 1417-1425 (2022).
10. Salah, H.M. & Mehta, J.L. Best time for administration of antihypertensive medications: morning or evening? Lancet (London, England) 400, 1383-1385 (2022).
11. Smithwick, R.H. & Thompson, J.E. Splanchnicectomy for essential hypertension; results in 1,266 cases. J Am Med Assoc 152, 1501-1504 (1953).
12. Symplicity, H.T.N.I. Catheter-based renal sympathetic denervation for resistant hypertension: durability of blood pressure reduction out to 24 months. Hypertension 57, 911-917 (2011).
13. Symplicity, H.T.N.I. et al. Renal sympathetic denervation in patients with treatment-resistant hypertension (The Symplicity HTN-2 Trial): a randomised controlled trial. Lancet (London, England) 376, 1903-1909 (2010).
14. Bhatt, D.L. et al. A controlled trial of renal denervation for resistant hypertension. The New England journal of medicine 370, 1393-1401 (2014).
15. Kandzari, D.E. et al. Effect of renal denervation on blood pressure in the presence of antihypertensive drugs: 6-month efficacy and safety results from the SPYRAL HTN-ON MED proof-of-concept randomised trial. Lancet (London, England) 391, 2346-2355 (2018).
16. Azizi, M. et al. Ultrasound renal denervation for hypertension resistant to a triple medication pill (RADIANCE-HTN TRIO): a randomised, multicentre, single-blind, sham-controlled trial. Lancet (London, England) 397, 2476-2486 (2021).
17. Bohm, M. et al. Efficacy of catheter-based renal denervation in the absence of antihypertensive medications (SPYRAL HTN-OFF MED Pivotal): a multicentre, randomised, sham-controlled trial. Lancet (London, England) 395, 1444-1451 (2020).
18. Townsend, R.R. et al. Catheter-based renal denervation in patients with uncontrolled hypertension in the absence of antihypertensive medications (SPYRAL HTN-OFF MED): a randomised, sham-controlled, proof-of-concept trial. Lancet (London, England) 390, 2160-2170 (2017).
19. Azizi, M. et al. Endovascular ultrasound renal denervation to treat hypertension (RADIANCE-HTN SOLO): a multicentre, international, single-blind, randomised, sham-controlled trial. Lancet (London, England) 391, 2335-2345 (2018).
20. Mahfoud, F. et al. Long-term efficacy and safety of renal denervation in the presence of antihypertensive drugs (SPYRAL HTN-ON MED): a randomised, sham-controlled trial. The Lancet 399, 1401-1410 (2022).
21. Sarathy, H. & Salman, L.A. Can renal denervation replace medications for patients with hypertension? Lancet (London, England) 399, 1363-1365 (2022).
22. Bhatt, D.L. et al. Long-term outcomes after catheter-based renal artery denervation for resistant hypertension: final follow-up of the randomised SYMPLICITY HTN-3 Trial. Lancet (London, England) 400, 1405-1416 (2022).
23. Mahfoud, F. et al. Cardiovascular Risk Reduction After Renal Denervation According to Time in Therapeutic Systolic Blood Pressure Range. Journal of the American College of Cardiology 80, 1871-1880 (2022).
24. Azizi, M. et al. Effects of Renal Denervation vs Sham in Resistant Hypertension After Medication Escalation: Prespecified Analysis at 6 Months of the RADIANCE-HTN TRIO Randomized Clinical Trial. JAMA cardiology 7, 1244-1252 (2022).
25. Noubiap, J.J. et al. Global prevalence of resistant hypertension: a meta-analysis of data from 3.2 million patients. Heart (British Cardiac Society) 105, 98-105 (2019).
26. Carey, R.M. et al. Resistant Hypertension: Detection, Evaluation, and Management: A Scientific Statement From the American Heart Association. Hypertension 72, e53-e90 (2018).
27. Imprialos, K.P. et al. Mineralocorticoid Receptor Antagonists in Essential and Resistant Hypertension. Curr Pharm Des 24, 5500-5507 (2018).
28. Williams, B. et al. Spironolactone versus placebo, bisoprolol, and doxazosin to determine the optimal treatment for drug-resistant hypertension (PATHWAY-2): a randomised, double-blind, crossover trial. Lancet (London, England) 386, 2059-2068 (2015).
29. Freeman, M.W., Bond, M., Murphy, B., Hui, J. & Isaacsohn, J. Results from a phase 1, randomized, double-blind, multiple ascending dose study characterizing the pharmacokinetics and demonstrating the safety and selectivity of the aldosterone synthase inhibitor baxdrostat in healthy volunteers. Hypertension research : official journal of the Japanese Society of Hypertension 46, 108-118 (2023).
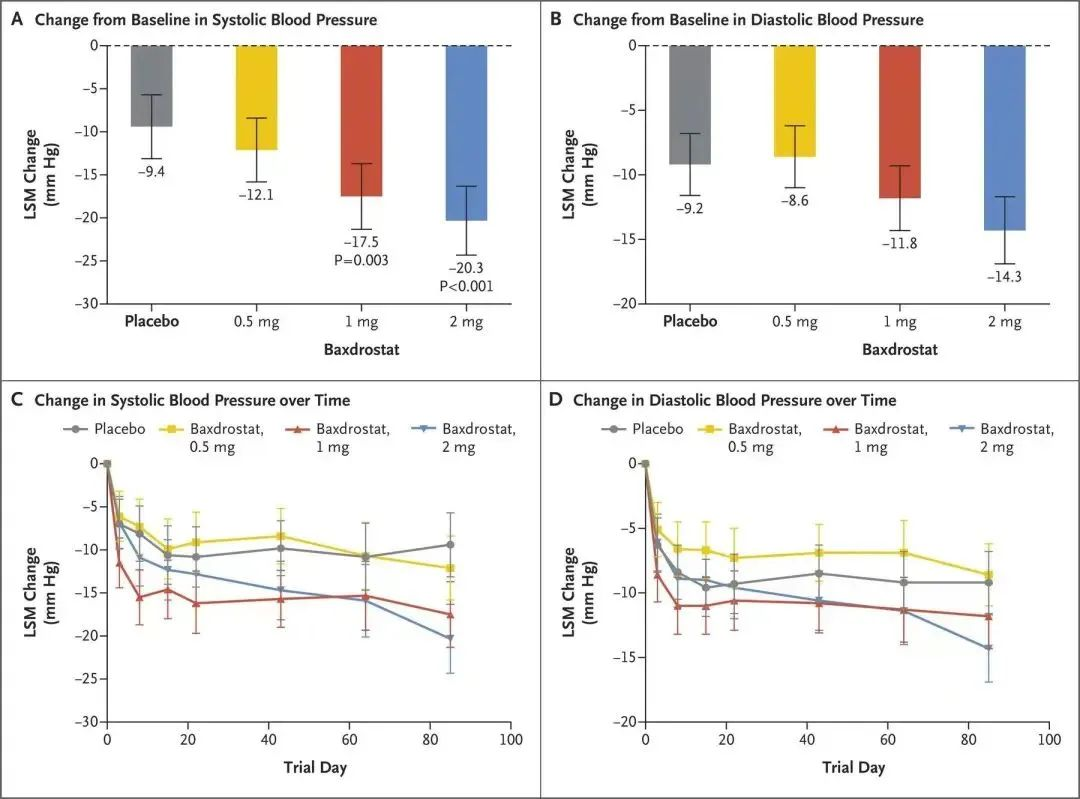
30. Freeman, M.W. et al. Phase 2 Trial of Baxdrostat for Treatment-Resistant Hypertension. The New England journal of medicine (2022).
31. Clozel, M. Aprocitentan and the endothelin system in resistant hypertension. Can J Physiol Pharmacol 100, 573-583 (2022).
32. Grubbs, A.L., Anstadt, M.P. & Ergul, A. Saphenous vein endothelin system expression and activity in African American patients. Arterioscler Thromb Vasc Biol 22, 1122-1127 (2002).
33. Rossignol, P. et al. The double challenge of resistant hypertension and chronic kidney disease. Lancet (London, England) 386, 1588-1598 (2015).
34. Solini, A. et al. Resistant hypertension in patients with type 2 diabetes: clinical correlates and association with complications. J Hypertens 32, 2401-2410; discussion 2410 (2014).
35. Krum, H., Viskoper, R.J., Lacourciere, Y., Budde, M. & Charlon, V. The effect of an endothelin-receptor antagonist, bosentan, on blood pressure in patients with essential hypertension. Bosentan Hypertension Investigators. The New England journal of medicine 338, 784-790 (1998).
36. Nakov, R., Pfarr, E., Eberle, S. & Investigators, H. Darusentan: an effective endothelinA receptor antagonist for treatment of hypertension. American journal of hypertension 15, 583-589 (2002).
37. Verweij, P., Danaietash, P., Flamion, B., Menard, J. & Bellet, M. Randomized Dose-Response Study of the New Dual Endothelin Receptor Antagonist Aprocitentan in Hypertension. Hypertension 75, 956-965 (2020).
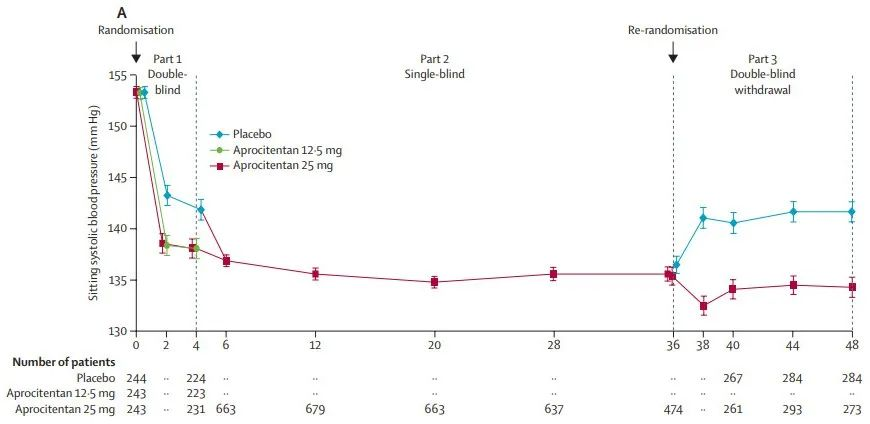
38. Schlaich, M.P. et al. Dual endothelin antagonist aprocitentan for resistant hypertension (PRECISION): a multicentre, blinded, randomised, parallel-group, phase 3 trial. Lancet (London, England) 400, 1927-1937 (2022).
39. Dechend, R. & Delles, C. Endothelin antagonists and the quest for a new therapeutic option in resistant hypertension. Lancet (London, England) 400, 1900-1901 (2022).
40. American College of, O. & Gynecologists' Committee on Practice, B.-O. ACOG Practice Bulletin No. 203: Chronic Hypertension in Pregnancy. Obstet Gynecol 133, e26-e50 (2019).
41. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists' Task Force on Hypertension in Pregnancy. Obstet Gynecol 122, 1122-1131 (2013).
42. Battarbee, A.N., Sinkey, R.G., Harper, L.M., Oparil, S. & Tita, A.T.N. Chronic hypertension in pregnancy. Am J Obstet Gynecol 222, 532-541 (2020).
43. Sinkey, R.G. et al. Prevention, Diagnosis, and Management of Hypertensive Disorders of Pregnancy: a Comparison of International Guidelines. Current hypertension reports 22, 66 (2020).
44. Garovic, V.D. et al. Hypertension in Pregnancy: Diagnosis, Blood Pressure Goals, and Pharmacotherapy: A Scientific Statement From the American Heart Association. Hypertension 79, e21-e41 (2022).
45. Sibai, B.M., Mabie, W.C., Shamsa, F., Villar, M.A. & Anderson, G.D. A comparison of no medication versus methyldopa or labetalol in chronic hypertension during pregnancy. Am J Obstet Gynecol 162, 960-966; discussion 966-967 (1990).
46. Magee, L.A. et al. Less-tight versus tight control of hypertension in pregnancy. The New England journal of medicine 372, 407-417 (2015).
47. Ankumah, N.A. et al. Risk of adverse pregnancy outcomes in women with mild chronic hypertension before 20 weeks of gestation. Obstet Gynecol 123, 966-972 (2014).
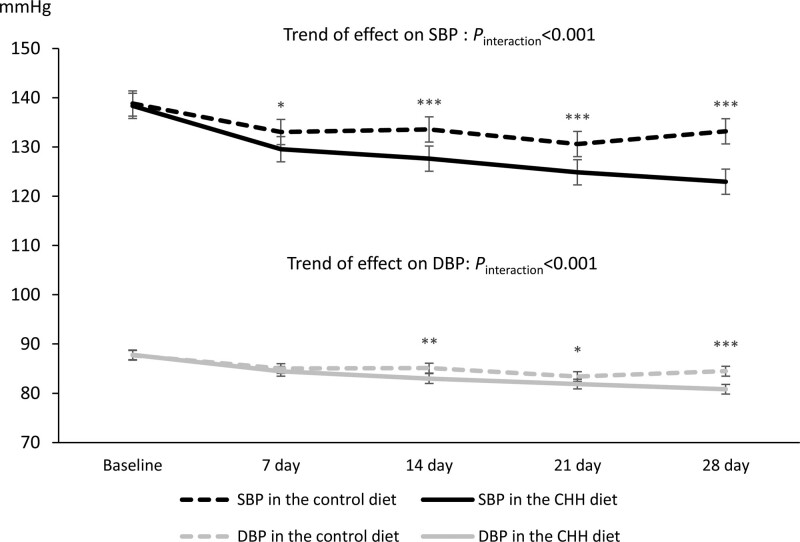
48. Tita, A.T. et al. Treatment for Mild Chronic Hypertension during Pregnancy. The New England journal of medicine 386, 1781-1792 (2022).
49. Huynh, K. Treatment of mild chronic hypertension in pregnancy reduces pre-eclampsia risk. Nature reviews. Cardiology 19, 350 (2022).
50. James, P.A. et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). Jama 311, 507-520 (2014).
51. Dorsch, M.P., Gillespie, B.W., Erickson, S.R., Bleske, B.E. & Weder, A.B. Chlorthalidone reduces cardiovascular events compared with hydrochlorothiazide: a retrospective cohort analysis. Hypertension 57, 689-694 (2011).
52. Roush, G.C., Holford, T.R. & Guddati, A.K. Chlorthalidone compared with hydrochlorothiazide in reducing cardiovascular events: systematic review and network meta-analyses. Hypertension 59, 1110-1117 (2012).
53. Whelton, P.K. et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Journal of the American College of Cardiology 71, e127-e248 (2018).
54. Hripcsak, G. et al. Comparison of Cardiovascular and Safety Outcomes of Chlorthalidone vs Hydrochlorothiazide to Treat Hypertension. JAMA Intern Med 180, 542-551 (2020).
55. Edwards, C. et al. Comparison of Clinical Outcomes and Safety Associated With Chlorthalidone vs Hydrochlorothiazide in Older Adults With Varying Levels of Kidney Function. JAMA Netw Open 4, e2123365 (2021).
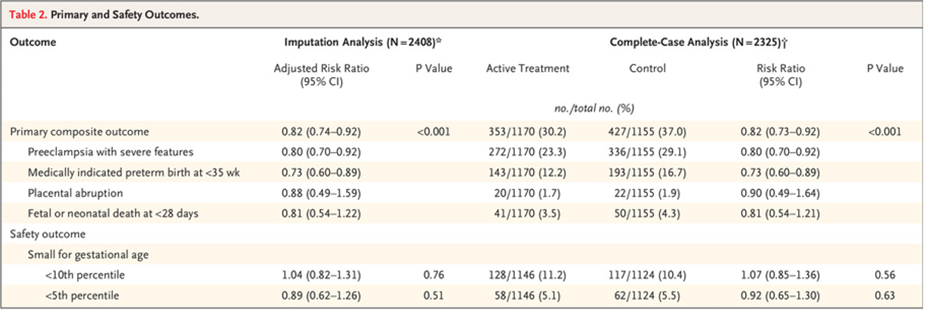
56. Ishani, A. et al. Chlorthalidone vs. Hydrochlorothiazide for Hypertension-Cardiovascular Events. The New England journal of medicine 387, 2401-2410 (2022).
57. Mills, K.T. et al. Global Disparities of Hypertension Prevalence and Control: A Systematic Analysis of Population-Based Studies From 90 Countries. Circulation 134, 441-450 (2016).
58. 马丽媛, 王增武, 樊静 & 胡盛寿 《中国心血管健康与疾病报告2021》关于中国高血压流行和防治现状 %J 中国全科医学. 25, 3715-3720 (2022).
59. Sidel, V.W. The barefoot doctors of the People's Republic of China. The New England journal of medicine 286, 1292-1300 (1972).
60. Li, X. et al. The primary health-care system in China. Lancet (London, England) 390, 2584-2594 (2017).
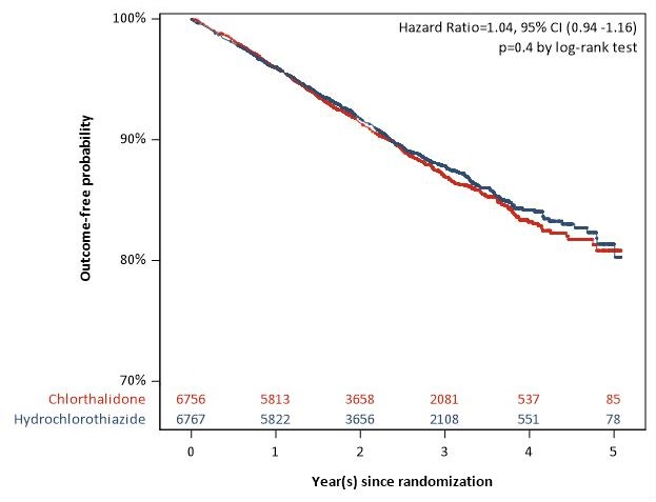
61. Sun, Y. et al. A village doctor-led multifaceted intervention for blood pressure control in rural China: an open, cluster randomised trial. Lancet (London, England) 399, 1964-1975 (2022).
62. Collaborators, G.B.D.D. Health effects of dietary risks in 195 countries, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet (London, England) 393, 1958-1972 (2019).
63. Wu, Y., Benjamin, E.J. & MacMahon, S. Prevention and Control of Cardiovascular Disease in the Rapidly Changing Economy of China. Circulation 133, 2545-2560 (2016).
64. Appel, L.J. et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. The New England journal of medicine 336, 1117-1124 (1997).
65. de Lorgeril, M. et al. Mediterranean diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: final report of the Lyon Diet Heart Study. Circulation 99, 779-785 (1999).
66. Estruch, R. et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. The New England journal of medicine 378, e34 (2018).
67. Obarzanek, E. et al. Effects on blood lipids of a blood pressure-lowering diet: the Dietary Approaches to Stop Hypertension (DASH) Trial. Am J Clin Nutr 74, 80-89 (2001).
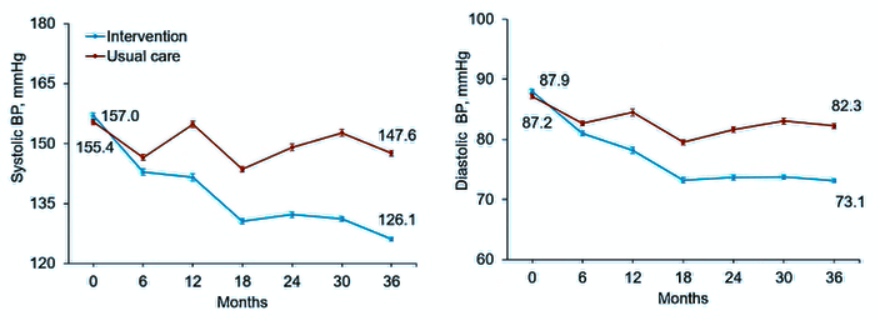
68. Wang, Y. et al. Effects of Cuisine-Based Chinese Heart-Healthy Diet in Lowering Blood Pressure Among Adults in China: Multicenter, Single-Blind, Randomized, Parallel Controlled Feeding Trial. Circulation 146, 303-315 (2022).
69. Bisognano, J.D. et al. Baroreflex activation therapy lowers blood pressure in patients with resistant hypertension: results from the double-blind, randomized, placebo-controlled rheos pivotal trial. Journal of the American College of Cardiology 58, 765-773 (2011).
70. Heusser, K. et al. Acute Response to Unilateral Unipolar Electrical Carotid Sinus Stimulation in Patients With Resistant Arterial Hypertension. Hypertension 67, 585-591 (2016).
71. Freeman, R. Clinical practice. Neurogenic orthostatic hypotension. The New England journal of medicine 358, 615-624 (2008).
72. Spiering, W. et al. Endovascular baroreflex amplification for resistant hypertension: a safety and proof-of-principle clinical study. Lancet (London, England) 390, 2655-2661 (2017).
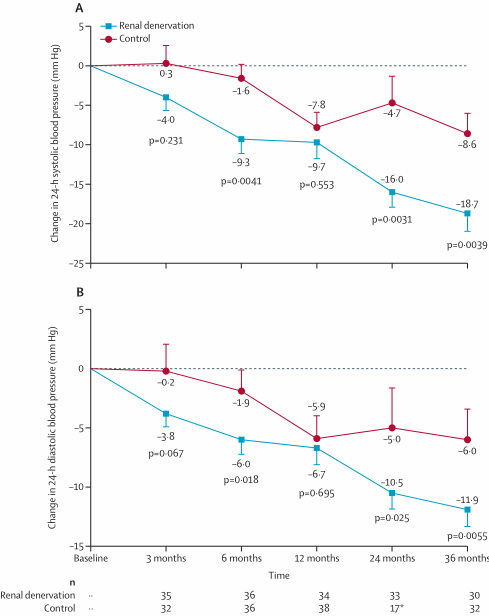
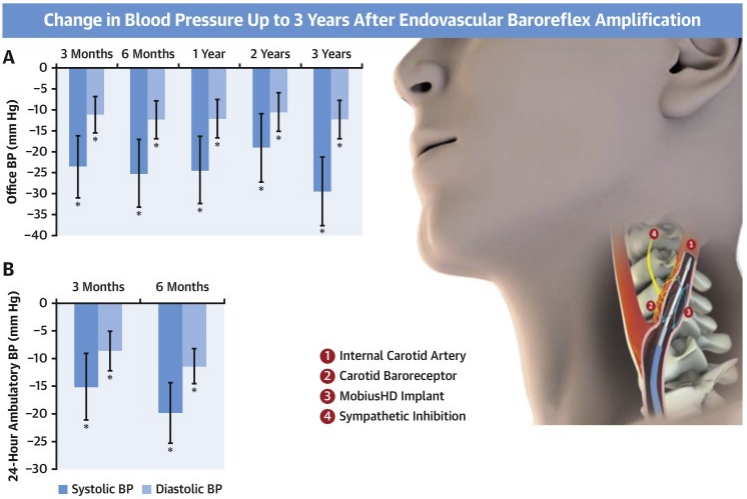
73. van Kleef, M. et al. Treatment of Resistant Hypertension With Endovascular Baroreflex Amplification: 3-Year Results From the CALM-FIM Study. JACC. Cardiovascular interventions 15, 321-332 (2022).
74. Maimaris, W. et al. The influence of health systems on hypertension awareness, treatment, and control: a systematic literature review. PLoS Med 10, e1001490 (2013).
76. China, P.C.G. et al. Primary care institutional characteristics associated with hypertension awareness, treatment, and control in the China PEACE-Million Persons Project and primary health-care survey: a cross-sectional study. Lancet Glob Health 11, e83-e94 (2023).